Dr. Parnell has been retained in medical device litigation cases that are in the news and in class actions. Products include Stents, Stent-Grafts, IVC Filters (Inferior Vena Cava Filters), Orthotics, Spinal Bracing, Surgical Bracing, Prosthetic Limbs, Orthopedic Devices, Intragastric Abdominal Balloon Weight Loss Systems, Back Pain Reduction Systems, Spinal Surgical Tools, and others.
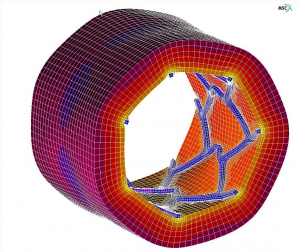
PEC has significant Medical Device experience including both implantable (PMA) and non-implantable (510K) components. We often apply nonlinear finite element analysis for design validation, design improvement, FDA regulatory submissions, and failure investigation.
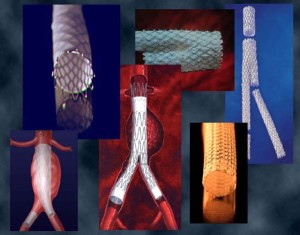
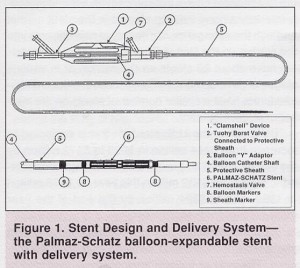
Some example applications from earlier projects (see figures below) include:
- Stents
- Stent-grafts
- Closure devices (arterial access for minimally invasive procedures; catheter access)
- Surgical Clips, Ligation Clips, Ligature (minimally invasive)
- Spinal devices; spinal surgical devices; spinal fixation
- Orthopedic devices, foot & ankle staples, plates
Materials include Nitinol (shape memory alloy), Stainless Steel, MP35N, Titanium, Elgiloy, various polymers, and plastics.
 |
 |
||
| CrossRoads Extremity Systems | |||
| Nitinol fixation devices for foot & ankle applications | |||